Preparation of Standard Solution of Sodium Carbonate - Chemistry

By A Mystery Man Writer
A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU
A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU'S to understand more about it.

Sodium Carbonate, 30 g

Standard solution, Resource

SOLUTION: Preparation of Sodium Carbonate by Solvay's Process

Titration of Sodium Carbonate With Hydrochloric Acid

Buffer solution pH 4 (phthalate), for pH measurement

Standard Solution

Standard Solution-Sodium Carbonate

Titration Lab Report - Ap0304 Practical Transferable Skills

Exp-27 Preparation of a standard solution of sodium carbonate
Transition Metals - Iodine-Sodium Thiosulfate Titrations (A-Level

Sodium carbonate - Wikipedia

Chp.13 - Volumetric analysis (acids/bases) [all] Flashcards
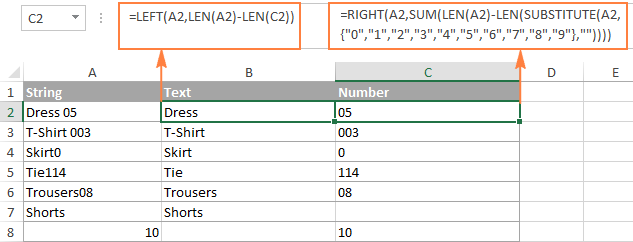
- Excel: Split string by delimiter or pattern, separate text and numbers
- Sector of a Circle - Area, Perimeter and Arc Length Formula

- How to determine the appropriate sample size for structural equation modeling - Statistics Solutions

- Diana Dressform - Size B - Dress Size 14-20 – Fabricville

- Use Online Shirt Size Calculator Find T Shirt Size with Height

- Printed Trousers at Cotton Traders

- Fesfesfes Teen Girls Cute Monokini Children Girls Back Hollow Out

- KaLI_store Mens Sweatpants Men's Drawstring Waist Side Pocket Straight Cargo Pants Joggers Khaki,3XL

- Pants & Jumpsuits, Lululemon Adapted State Jogger Size 2

- Dominique Women's Jillian Wire-free Unlined Minimizer Bra - 6800 42d Pink : Target
