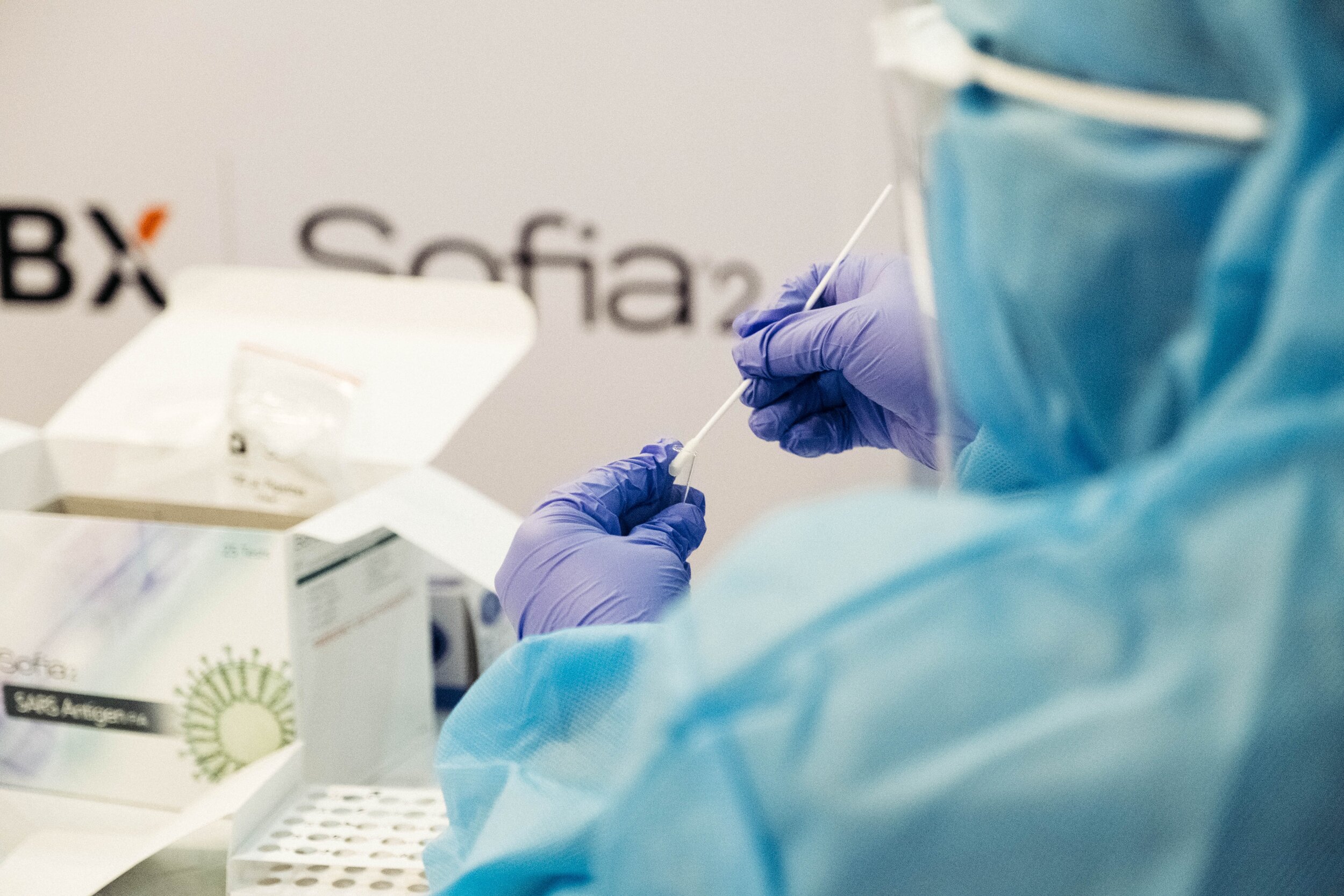
Crucial Steps for Singapore Medical Device Registration & HSA Approval

By A Mystery Man Writer
Discover the crucial steps for successful Singapore medical device registration and HSA approval. Operon Strategist offers expert guidance, classification insights, and comprehensive support. Contact Operon Strategist to learn more and navigate the regulatory landscape with confidence.

Start a Pharmaceutical Company in Singapore in 2023: Steps & License Requirements

Identifying Predicate Devices in Asian Countries

Medical Device Registration in Singapore – Pre-submission Meetings with Health Sciences Authority

Crucial Steps for Singapore Medical Device Registration & HSA Approval

HSA Revised Guidance on Medical Device Product Registration: Class A and B

EXCLUSIVE! Revolutionising Healthcare: Unlocking the Future with Technology – OpenGov Asia

Singapore tweaks rules to fast-track product access

Regulatory Oversight of Cell, Tissue, and Gene Therapy Products in Singapore

Post-approval of CMC changes under FDA, EMA and HSA guidance - No deviation
- Unbranded 00 Style Diamond Tights

- Generic Cotton Ion Lifting Lymphvity Detoxification Bra Nylon Ion Shaping Bra New Design Tourmaline Slimming Bra For Women Yoga Sports @ Best Price Online
/product/60/7982671/1.jpg?5069)
- 50 Popular French Crop Haircuts For Men To Copy in 2024

- Starry Beaded Navy Blue Premium Cotton Shirt – The Foomer

- How Marilyn Monroe and Audrey Hepburn Represented Two Types of Femininity to Hollywood